|
That’s more than twice the number of rules as 1blocker legacy with room to grow to 350,000 rules in the future.ġBlocker X introduces a new set of blockers that legacy version didn’t have, which is regional blockers. Group of blockers that removes ads from most sites.īlocks sites from tracking you across the web.īlocks social media widgets, cookie notices, share bars, crypto-miners, etc.īlock comments on many popular sites where they aren’t the primary focus. The blockers you activate are synced to all your iOS devices using iCloud Safari settingsġBlocker X includes about 120,000 rules organized in the following categories: Once enabled, you can turn each of the 7 sets of rules or individual rules on and off from within 1Blocker X. In Safari’s settings, there’s a Content Blockers section where you can turn on 1Blocker X’s 7 blockers. 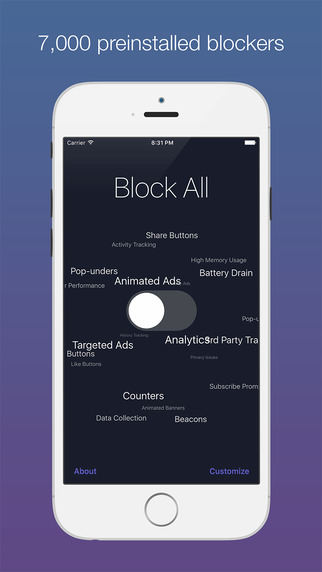
To get going, you have to enable 1Blocker’s content blockers by visiting the Settings app. This required rewriting the app to handle new extensions and thats why there is a new app called 1blocker X and that also is the reason why this is a paid upgrade. To get over the limit of 50,000 blocking rules in the app, 1blocker team came up with the idea of creating multiple extensions within safari that independently manage blocking. Developers of 1blocker wanted to get over that limit of 50,000 rules and block more unwanted content this lead to the development of 1blocker X that is replacing the old 1blocker app. That’s a lot of rules, but they are not enough considering the amount of spam on the internet. I paid for its IAP to unlock the full app, it had about 50,000 rules, as iOS limits the number of blocking rules that can be implemented by an app to 50,000. It is advised that such input be obtained prior to moving forward, to make sure that all data generated in any further work contributes to the design and execution of the next steps, and also contributes to a development/regulatory perspective in case of a positive outcome.I have been a user of 1blocker for iOS, for about 2 years. Overall, TACT advise that the applicant recruits a preclinical and regulatory expert advisory committee to address their preclinical work-package status and planning, and to develop an adequate dossier for obtaining scientific advice, guidance and regulatory input from the regulatory authorities. Peacock Pharma are planning additional preclinical work and are developing plans for the First In Man (FIM) study in healthy adult volunteers, and a short duration (14 days) pilot phase 2A study in 15 adult Becker muscular dystrophy (BMD) patients. Nevertheless, this project is at a relatively early stage of development and the preclinical package is not fully complete. These data indicate a possible beneficial effect of KR-33028 on intracellular calcium uptake and grip test performance. The applicant has generated some encouraging but limited preclinical pilot data with KR-33028 in the mdx mouse model. Consequently, inhibiting NHE-1 may reduce calcium overload and improve muscular function in patients with muscular dystrophies. Calcium overload of skeletal and cardiac muscles is a key contributor to the pathophysiology of muscular dystrophies, and overactivation of NHE-1 may contribute to calcium overload. Peacock Pharma are investigating the use of a selective NHE-1 (Na/H-exchanger-1) blocker, KR-33028, as a potential therapeutic for the treatment of dystrophin-deficient muscular dystrophies. Stem cell therapy for SMA Type1 Children.Early access to medicines in development.Congenital Muscle Disease International Registry.Family Registry for Centronuclear and Myotubular Myopathies.Best practice guidelines for molecular testing published in Neuromuscular Disorders.Accredited laboratories for genetic diagnosis.Standards of Care for Congenital Myopathy (CM).The Guide to the 2017 International Standards of Care for SMA.Ongoing updating and dissemination of standards of care for DMD.Creation of the DMD care standards document.The Diagnosis & Management of DMD in different languages.NMR (MRI and MRS) as an outcome measure.Overview of therapeutic approaches for SMA.Facing the Challenges of Clinical Trials.Common protocols in MDC1A preclinical research.Experimental protocols for SMA animal models.Experimental protocols for DMD animal models.TREAT-NMD Post Marketing Surveillance Webinar.Post Marketing Surveillance Frequently Asked Questions.TREAT-NMD Global Data systems Oversight Committee – TGDOC.Myotubular and Centronuclear Myopathies.Limb Girdle Muscular Dystrophies (LGMD) Core Dataset.Facioscapulohumeral Muscular Dystrophy (FSHD) Core Dataset.Duchenne Muscular Dystrophy (DMD) Core Dataset.Spinal Muscular Atrophy (SMA) Core Dataset.TREAT-NMD Education Committee Endorsement.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
